Thursday, December 4, 2014
Presentation Week
Well the semester is coming to an end and I am not ready stop! This semester has been amazing. I learned so much working on this project. Although my final paper has been turned in I still have a few more tests that I would like to complete before this semester finally ends. I know know that proteins can be extracted from tissue samples, and that antibodies can be extracted from these proteins. I know that sheep brains can contain the beta amyloid proteins that are responsible for Alzheimers Disease (AD). Today I found a study that was performed at a university in Japan that was super empowering. They successfully measured the Beta amyloid proteins from individuals receiving treatment for AD. Now, all that is required is to put this all together and continue to work on my technique. Hopefully next semester will yield a tangible test that can be used in everyday application. However, my secondary hypothesis has proven to most challenging (Is it possible to consume an entire bag of tasty oh's in a semester?)

Thursday, November 20, 2014
Just Keep Swimming!
This week has been absolutely crazy for me. I had three exams and final project deadlines are quickly approaching! This week in lab I have worked on trying to come up with an idea for obtaining brain slices that are thin enough for me to see proteins but strong enough to survive the staining process. I started by selecting three brains that are positive for frontal lobe hematoma and three brains that do not have any visible hematoma. I am not sure that there is any significance to this but, I am curious as to why approximately 40 percent of the brains in an unopened bucket are positive and the rest are not. I then used a coring instrument to obtain three samples from each brain and placed them in the -80 degree freezer for 24 hours. The coring process was not much fun, the preservative smell and the close proximity needed for an accurate sample left me feeling a bit nauseous. Perhaps I will do only three at a time for my next coring session. Next I very slowly made slices using my makeshift microtome. I then took these slices and stained them using crystal violet. I have been able to slice them extremely thin but once I start staining them they disintegrate, littering the sink with brain matter which is a bit gross. The problem with such thin slices is that they thaw once they hit the room temperature microscope slides. Perhaps freezing them again before staining along with the microscope slides will help? I also got to spend time discussing projects with some of my S-STEM colleagues, celebrated Matt Hills birthday, and tried to offer a warm welcome to our newest STEMmers who were busy with their unknown microbe projects. All in all, it was a great week and I am excited to see everyone's presentations in the very near future!
| Hematoma Brain |
| Normal Brain |
Thursday, November 13, 2014
S-STEM Field trip
Last Friday I had the good fortune of my first S-STEM field trip. We went to Dreamy Draw park and I got to hike and experience geocaching for the very first time with some pretty amazing people. Thank you to our fearless (and extremely knowledgeable) tour guides Matt and Josh. I learned about femoral drainage and the mining of mercury that used to take place there. It was a much needed break from this stressful semester, and I am so happy that not only has S-STEM been an amazing experience, but I did not realize the bonds that would be formed from spending so much time together in the lab.
| This is a pic of Matt and I attempting microtome #2 |
Thursday, November 6, 2014
The search for a new brain
This week has been an exciting eek for me in the lab. I have successfully created a microtome! Ok, it is a very archaic design, but functional nonetheless. I used a 20 cc syringe, a brass corer (size 7), and a blade from a box cutter. The tip of the syringe was removed using a box cutter. After much measurement I realized that the diameter thickness that I wish to acieve just happens to be the distance between the cc markings on the syringe. Matt Hill used a saw to create an opening for my blade. the brain is cored and placed into the syringe. a smaller corer is used to plunge the brain down the syringe until it is pressed against the blade resting in its position within the cut in the syringe. when the blade is released the brain is moved ever so slowly forward and the blade slices through it. This little device has created some adequate samples for staining (using crystal violet) so I am very happy with it. However, the lego microtome is still in the process of being built (thanks to Matt Hill) but is not quite finished yet. The first brain that I sectioned was chosen due to the presence of a hematoma on the frontal lobe and a decreased frontal lobe size. This brain contained proteins when stained and viewed under the microscope. However, frozen brain tissue is much easier to cut so I placed it in the -80 degree freezer so that it would be super frozen. Well, when I applied the pressure to it in order to core it...it exploded into pieces which flew all over the bench and floor. although impressive it was really unpleasant to clean up (it defrosts quickly in such small pieces.) Now brainless, I picked another brain randomly from the bucket. after getting a sample under the microscope I quickly realized that it did not contain the proteins that I was looking for. I chose another brain randomly and again obtained negative results. I began searching the buckets for a third brain vowing to be picky this time to find one that shared the same characteristics as the first brain. I found the perfect specimen at the bottom of the bucket. Unfortunately I ended up dipping my leather banded watch in the process of retrieving it, which my fellow STEMmer Jeremy found extremely entertaining. This brain is positive for proteins. I am not sure if this is coincidence or not but it is defiantly interesting.
http://www.google.com/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0CAYQjB0&url=http%3A%2F%2Fwww.scbt.com%2Fdatasheet-53822-beta-amyloid-20-1-antibody.html&ei=Kk9cVNP1MY72iQLCn4G4AQ&bvm=bv.79189006,d.cGE&psig=AFQjCNGoJW91fjtiLzgLVGozUWqAPsl6bA&ust=1415422087904595
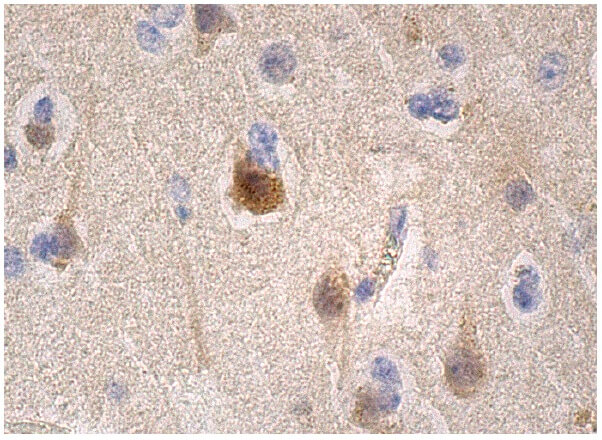 |
| Beta amyloid present in brain tissue. |
http://www.google.com/url?sa=i&rct=j&q=&esrc=s&source=images&cd=&cad=rja&uact=8&ved=0CAYQjB0&url=http%3A%2F%2Fwww.scbt.com%2Fdatasheet-53822-beta-amyloid-20-1-antibody.html&ei=Kk9cVNP1MY72iQLCn4G4AQ&bvm=bv.79189006,d.cGE&psig=AFQjCNGoJW91fjtiLzgLVGozUWqAPsl6bA&ust=1415422087904595
Thursday, October 30, 2014
Brains, Stains, and Pains
So this week was filled with good news and bad news. The bad news is that my western blot was not successful, however I did learn a ton from just completing the process. The good news is that I have completed collecting data for this part of my project and I am confident that I can extract the amyloid proteins should I be lucky enough to find a sample that contains them. I read an interesting article pertaining to these specific proteins being present in the brains of sheep. This is super lucky for me due to the abundance of them in our lab. I have read many articles related to staining proteins and which stains work best. It seems that; crystal violet, Iodine, and comaisse blue seen to be the most popular. I prepared brain samples on slides and used each stain just to see if one was easier to interpret than the others ( and because I was super curious). I liked the crystal violet best but my samples were too thick. A microtome would make this step so much easier but they are pretty expensive (although a lego version is currently under construction thanks to Matt H and Josh). I need each sample to be no thicker than 250 nm. Stains have been ordered, so next week promises much fun!

Thursday, October 23, 2014
This Weeks Post is Brought to you by the Letter J
Josh, J-E-L-L-O, jousting....three words that describe this week in the lab. Protein extraction through electrophoresis was not something that I was not familiar with before this semester. I successfully cut up my frozen fish samples into 0.25cm, watched C3PO add DDT to it (its harmful if inhaled), placed it in the water bath for 5 minutes at 95 degrees Celsius, loaded my gel (which comes pre-assembled in a very hard plastic cassette, added buffer to the tank, set the volts at 200, and crossed my fingers for about an hour. After the cycle was complete I rinsed the gel cassette and realized that this particular cassette requires a "key" which is not included, but can be purchased for $5.00 (plush shipping/handling). Let's Just say that I was not pleased with the manufacturers of this particular product. I had spent hours on this particular gel and was not going to lose my results because of a silly "key". I did some quick investigating of the mechanics of this cassette and decided that the most rational approach would be to just break it. I began by using the plastic comb that was removed from the wells of the gel but the cassette was not budging and was not giving up. Luckily Josh (the biker scout), sensing my frustration at Jousting with this cassette, came to the rescue with an extremely large flat head screwdriver. I quickly accepted this gift, had a fleeting thought of smashing the cassette with the tool, and finally decided to take his advice and use it to pry the thing apart. After a few minutes the cassette acquiesced and released my beloved gel. After it was rinsed and stained (thanks again to Josh for help with this process) I recorded my results. I was left with a gel full of data that was going to start to degrade which gave me a very sad feeling. I was not ready to part with this little masterpiece so I immediately went online to find out how others have successfully handled this situation. The answer was immediate and brilliant.....J-E-L-L-O! Gels can be preserved in a 90/10 Jello solution for long lengths of time. Now....on to Western Blotting!!
| |
Thursday, October 9, 2014
One Fish, Two Fish....
This week I started the protein extraction process from five different
fish species. Biker Scout provided me with salmon and swai (which I have
never heard of) and we had a lively conversation of whether or not
canned tuna fish would work for this project (the answer was no, but we
both decided that it would be fun to try later). C3PO came to the rescue (with the help of the lab-mobile and some mechanics provided by our biker scout) and
provided very aromatic samples of catfish, dover, and halibut (which everyone else in the lab appreciated).. I am
very curious to find out how successful this technique is; for I would
like to try more evolved organisms after I master the fish. I also spent
some time swabbing a fellow stemmer which turned out to very amusing
and fun after the third hour. My fish have been cut up into 2.5 cm3
pieces and are marinating in buffer solution as I type this. My hope is that I will have results for my next post.


Tuesday, September 30, 2014
Electrophoresis V.S Western Blotting for detection of a protein
So after some intense dialog with the Biker Scout and C-3PO I think that I may be a bit closer to a project idea, even though it seems very far away from my goal upon first glance. This semester I will be using electrophoresis to isolate a specific protein. I will then use western blotting to introduce antibodies to the protein. Once I become fluent in this process my hope is to actually go after more specific human proteins/antibodies. I am curious to know if the antibodies produced from the beta amyloid protein could be used to create a test that would show the varying degree of beta amyloid proteins if they were present in an individual's blood. The first question that I had was; "Can beta-amyloid proteins be detected/isolated from urine and/or blood"? The answer is yes, they can be detected in blood, cerebral spinal fluid, and urine. My next question was,"Why is there not an inexpensive urinalysis (the least invasive procedure for the patient) that could detect the amount of beta amyloid proteins excreted through urine"? If the theory of the accumulation of these proteins or plaques is what causes the cognitive decline associated in Alzheimer's Disease is correct, than why are we not incorporating a device that will detect these plaques BEFORE they have time to build up? Many health care professionals already incorporate urinalysis into their annual physical protocol, why are we not monitoring the production of these proteins? Currently, the testing for these proteins is extremely costly and is almost always done on individuals that are ALREADY experiencing cognitive decline. I guess what I am asking is; when will we start to become proactive and get ahead of this disease, instead of ignoring it until it takes up space in our heads?


Thursday, September 25, 2014
Change in Plan
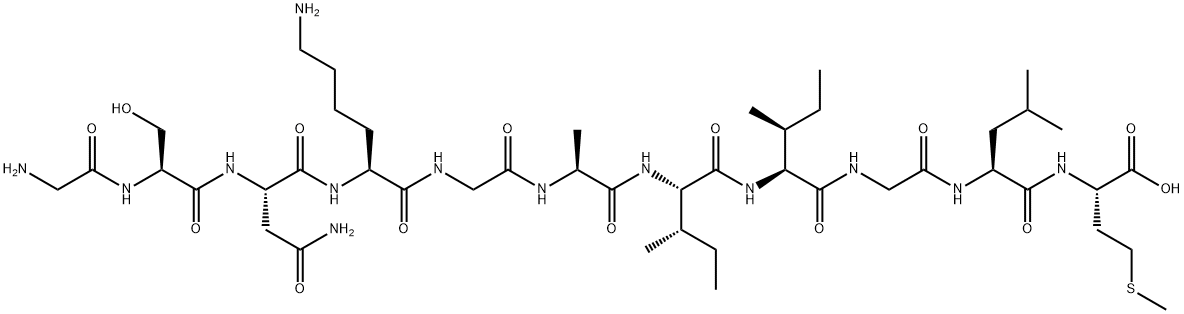
So apparently FISHing is super expensive, so I will not be doing it this semester. So with a heavy heart I will have to choose another project. I have always been curious about the beta amyloid plaques that accumulate in the brains of individuals affected with Alzheimer's Disease. These plaques are actually a sequence of 55 amino acids that most every person produces. The ability to excrete these proteins through urine reduces the probability of one being affected by this disease. When a person's body does not excrete the beta amyloid proteins they start to build up, like plaque on teeth. The once happy neurons frolicking happily in the brain are now plagued by these protein formations that prevent communication between one another. It takes many years for these formations to effect cognition which is why Alzheimer disease is usually reserved for the elderly population. I would like to see if I can isolate protein from blood in order to eventually acquire an antibody that I might study. If this project is successful, I would like to try to isolate beta amyloid protein from human blood. I wonder if I could start this process with blood from another organism?
Wednesday, September 17, 2014
What is the Difference Between FISH and a Karyotype?
FISH analysis will tell you if a specific chromosome is present and how many chromosomes there are and is much faster than a karyotype. FISH is used commonly for diagnosing down syndrome during pregnancy using amniotic fluid. Downs syndrome is diagnosed when there is a trisonomy on chromosome #21 (that is a number sign not a hash-tag). Now, what it will not tell you is the structural composition of the chromosome (like a karyotype) which is equally as important as knowing how many chromosomes are present. FISH analysis can take from 7-10 days. A karyotype can take up to two weeks to yield results. So, they are similar but very different procedures. Comparing FISHing to Karyotyping (not sure if either of those words are real words but you understand me for all intents and purposes) is like comparing an X-ray to an MRI. The X-ray is great for confirming the general location of pain that a patient is experiencing, but an MRI may be needed if there are further questions regarding structural integrity.So this semester I will learn to FISH, and if it is fun maybe I will try to karyotype (not myself though because I do not need to know that much about myself. It is already hard enough to compartmentalize the information I already have) totally kidding (kind of) :)
 |
| FISH (property of Breen lab at NCSU) |
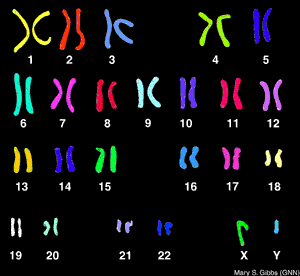 | ||
| KARYOTYPE |
Tuesday, September 16, 2014
FISHing
Hi there, this is my first ever blog so please keep that in mind while reading. I guess I will start with the first two questions that I had when I learned about FISH; what is FISH and why do I care? Fluorescence in situ hybridization (FISH) is a laboratory technique for detecting and locating a specific DNA sequence on a chromosome. The technique relies on exposing chromosomes to a small DNA sequence called a probe that has a fluorescent molecule attached to it. The probe sequence binds to its corresponding sequence on the chromosome.(http://ghr.nlm.nih.gov/glossar y=fluorescentinsituhyb ridization). FISH is used by geneticists in order to diagnose abnormalities within chomosomes, which can save lives. That is why I care. I am a little nervous about attempting this project because it takes extreme precision and requires that measurements be conducted at specific times. I am super excited and ready to get started!

© 2005 Nature Publishing Group Speicher, M. R. et al. The new cytogenetics: blurring the boundaries with molecular biology. Nature Reviews Genetics 6, 784 (2005). All rights reserved.
Subscribe to:
Comments (Atom)
 Figure 1: Principles of fluorescence in situ hybridization (FISH).
Figure 1: Principles of fluorescence in situ hybridization (FISH).